 Note that the majority of the plots for metals slope upward because both the metal and the oxide exist as condensed solid or liquid phases. Temperatures where either the metal or oxide melt (M) or vaporize (B) are marked on the diagram. Therefore, the diagram is drawn with DG=0 at the top of the diagram, and the values of DG shown are all negative numbers. The free energy of formation is negative for most metal oxides, which means the reaction can proceed without further influence. In a heat-treat system this is (for the most part) irrelevant, except in the brazing and sintering processes where those phase changes (melting) can indeed occur. The slopes of those plotted lines change when any of the materials involved melt (M) or vaporize/boil (B). Unless a phase change occurs, the free energy ( DG) versus temperature plot can be drawn as a series of straight lines, where DS is the slope and DH is the y-intercept. Since any explanation of how to calculate and plot an Ellingham diagram is more about the mechanics of the derivation process and less about how to use the tool, which is the intent of this article, the concept is covered very briefly here.Įnthalpy ( DH) and entropy ( DS) are essentially constant with temperature. The Ellingham diagram shown is for metals reacting to form oxides. 1 for the classic Ellingham diagram that would typically be used for heat-treating atmosphere processes.Īn Ellingham diagram is a plot of DG (change in Gibbs free energy) versus temperature, which, for our purposes, would be a temperature in a continuous furnace by zone or in a batch furnace by time in the cycle. We can adapt the diagram from its original use as a higher-temperature extractive-metallurgy tool to one where we can predict the effects of protective atmospheres and common atmosphere impurities and their impact on heat-treated product. Let’s restate and go stepwise through the Ellingham diagram to set up and make clearer how metal heat treaters can use it. The reduction of metal oxides with carbon (or carbon monoxide) to form the free, reduced metals is of immense industrial importance (blast furnace reduction of iron ores), and Ellingham diagrams show the lowest temperature at which the reaction will occur for each metal. In particular, and this reaction is critical to metal reduction systems that use carbon dioxide, he could show graphically that carbon becomes a stronger reducing agent as the temperature increases. His diagram and its variants help to select the best reducing agent for various ores in the extractive-metal process.Įllingham discovered that by normalizing the thermodynamic functions to a given reaction with one mole of oxygen he was able to compare the temperature stability of many different oxides on the same diagram. This phenomenon was known before Ellingham’s time, but Ellingham demonstrated it more clearly and made it more accessible to industry as a tool. The oxide plot tends to be the most common and will be highlighted here as its applicability to heat-treating processes is the most relevant. Harold Johann Thomas Ellingham (1897-1975) was a British physical chemist and is best known for the diagrams named after him that plot the change in standard free energy with respect to temperature for reactions like the formation of oxides, sulfides and chlorine of various elements, such as: This article demonstrates its usefulness. 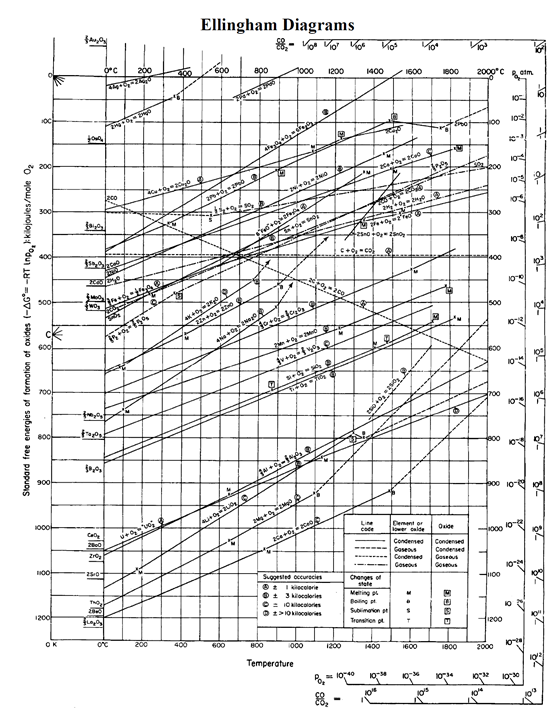
Thus the bronze age preceded the iron age the latter had to await the development of technology capable of producing higher temperatures, such as the blast furnace.The Ellingham diagram is not always thought of as a heat-treater’s tool. \) illustrates why the metals known to the ancients were mainly those such as copper and lead, which can be obtained by smelting at the relatively low temperatures that were obtainable by the methods available at the time in which a charcoal fire supplied both the heat and the carbon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
